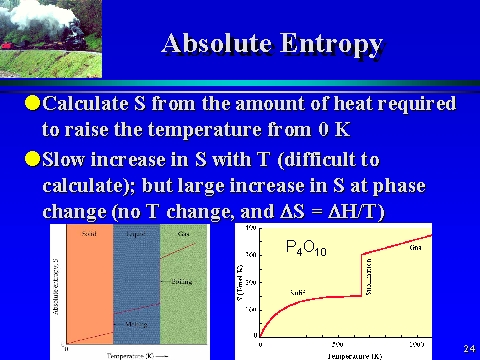
2 It is then convenient ('convenient' in the absence of further information) to take that common value to be zero: S (0) = 0. The third law of thermodynamics is an assertion about the unattainability of absolute zero, but it implies that the entropies of all pure, perfectly crystalline substances are the same at T = 0. Note that 'rev' no longer appears, so this expression applies regardless of the nature of the change in the system (it applies to both reversible and irreversible processes). Where q surr is the energy transferred as heat into the surroundings. Therefore, the change in entropy of the surroundings Δ S surr is calculated from: Because the surroundings are so huge and of infinite heat capacity, any change taking place in the system results in a transfer of energy as heat to the surroundings that is effectively reversible and isothermal. The expressions in (4) apply to the system. Enthalpies of transition, Δ trs H, are molar quantities (units: J mol -1, and typically kJ mol -1), so an entropy of transition has the units joules per kelvin per mole (J K -1 mol -1). Moreover, the expression applies only to the change in entropy at the transition temperature, for only then is the transfer of energy as heat a reversible process. In the third expression, note the modern location of the subscript denoting the type of transition (Δ trs X, not Δ X trs). If the volume is constant, use the isochoric heat capacity, the heat capacity at constant volume, C V. If the pressure is constant during heating, use the isobaric heat capacity, the heat capacity at constant pressure, C p. In the second expression the heat capacity C may be expressed in terms of the molar heat capacity C m (units: J K -1 mol -1), as C = nC m. In the first expression, n is the amount of gas molecules (units: mol) and R is the gas constant (units: J K -1 mol -1), which is defined in terms of Boltzmann's constant k (units: J K -1) and Avogadro's constant N A (units: mol -1) by R = N A k. Three applications of the above expressions are as follows: 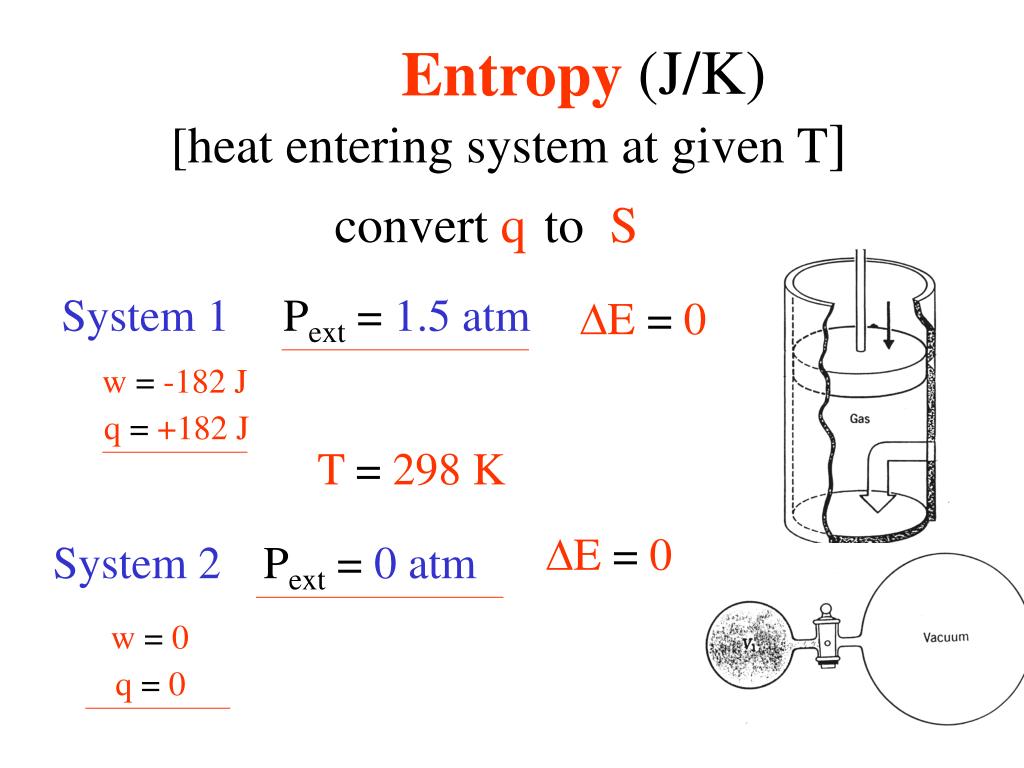
In practice, the external pressure or temperature needs to be infinitesimally different from that of the system to ensure the appropriate direction of change. In reversible heating, the temperature of the external heater is matched to the changing temperature of the system at all stages of the heating: there is thermal equilibrium throughout. In a reversible expansion, the external pressure is matched to the changing pressure of the system at all stages of the expansion: there is mechanical equilibrium throughout. In a reversible process, the system and its surroundings are in equilibrium. 
A reversible process is one that changes direction when an external variable, such as pressure or temperature, is changed by an infinitesimal amount. 
The definition refers to the reversible transfer of energy as heat. It is commonly demonstrated that S is indeed a state function by using the Carnot cycle, as explained in textbooks. It is far from obvious that the definition in (1) implies that S is a state function, or equivalently that d S is an exact differential (one with a definite integral that is independent of the path of integration). 1Įntropy is a state function that is, it has a value that depends only upon the current state of the system and is independent of how that state was prepared. With energy (and, by implication, heat) in joules (J) and temperature in kelvins (K), the units of entropy are joules per kelvin (J K -1). Where q rev is the total energy transferred reversibly during the change of state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
